HOCl molecular structure and oxidation-reduction potential (ORP) — operating at 850–1,100 mV, far exceeding the 650 mV microbial kill threshold
Why Hypochlorous Acid Is Transforming Wound Care
The science behind HOCl’s ability to eliminate pathogens, protect healthy tissue, and actively support the biology of healing — explained for everyone.
Curativa Bay Clinical Science Series · EPA Certified · FDA Recognized · WHO Cited
THE SCIENCE
Your Immune System Already Makes This
Hypochlorous acid (HOCl) is not a new invention. It is a molecule your own body produces every time it encounters a threat.
When bacteria or viruses enter the body, white blood cells called neutrophils respond immediately. Through a process called the myeloperoxidase pathway, these cells generate HOCl on-site — deploying it directly against the pathogen. This has been happening inside every human body since our immune systems evolved.
What scientists have spent decades learning to do is stabilize HOCl outside the body — in a form pure and consistent enough to be used clinically. The result is a wound care agent that works with the body’s own biology rather than against it, in a way no synthetic antiseptic can replicate.
| H₂O₂ + Cl⁻ → HOCl + H₂OMyeloperoxidase pathway — the neutrophil oxidative burst that generates HOCl during innate immune defense |
The chemistry matters because it explains everything else. HOCl is a neutral, protonated molecule. That neutrality gives it a property no other common antiseptic has: it can pass freely through microbial cell membranes, reaching its targets from the inside. Bleach (sodium hypochlorite) cannot do this — its negatively charged hypochlorite ion is repelled by the same charge on cell membranes, which is why HOCl is up to 80–100 times more antimicrobially effective at equivalent concentrations.
| HOCl does not merely disinfect surfaces. It penetrates microbial cells, disrupts their fundamental biology, and destroys them from within — just as your immune system intended. |
| 850–1,100 mVOperating ORP — far above 650 mV kill threshold | 80–100×More antimicrobial than bleach at neutral pH | < 30 secPathogen kill time across bacteria, viruses, fungi |
UNDERSTANDING ORP
Oxidation-Reduction Potential: The Measure That Actually Matters
You may have seen ORP mentioned on the Curativa Bay label and wondered what it means. Here is the plain-language version.
Oxidation-reduction potential (ORP) is a measurement — in millivolts — of how powerfully a solution can oxidize, meaning how aggressively it can strip electrons from other molecules. Microbial cells are vulnerable to oxidation. When a solution with high enough ORP contacts a pathogen, it essentially dismantles the pathogen’s cell structures at the chemical level.
The threshold for reliable microbial inactivation is approximately 650 mV. Chlorinated tap water sits around that level. Stabilized HOCl operates at 850–1,100 mV — substantially and consistently above the kill threshold. This is why HOCl achieves pathogen inactivation in under 30 seconds across bacteria, viruses, fungi, and even bacterial spores.
| Why conventional solutions fall short at the ORP level:• Drinking water: 200–400 mV — insufficient for reliable microbial inactivation• Seawater: ~400 mV — antimicrobially limited• Chlorinated water: ~650 mV — at the lower edge of the kill threshold• Stabilized HOCl: 850–1,100 mV — consistently, substantially above threshold |
But high ORP alone would not make HOCl special. Many chemical oxidizers have high ORP. What makes HOCl clinically significant is that it achieves this oxidizing power while remaining non-toxic to human tissue. Bleach is cytotoxic at wound concentrations. Hydrogen peroxide damages fibroblasts — the very cells that build new tissue. Chlorhexidine inhibits the cellular migration needed for wound closure. HOCl does none of these things.
| The critical insight is not just that HOCl kills pathogens effectively. It is that it does so without damaging the tissue that needs to heal. |
MECHANISMS OF ACTION
Four Ways HOCl Destroys Pathogens

HOCl operates through four simultaneous oxidative pathways — producing rapid, multi-target pathogen inactivation that prevents resistance development.
One of the reasons HOCl is so effective — and why pathogens cannot easily develop resistance to it — is that it does not rely on a single mechanism. It attacks through four distinct oxidative pathways simultaneously.
1. Membrane Oxidation
The outer membrane of a bacterial cell is made of phospholipids — a bilayer of fatty molecules that acts as the cell’s protective boundary. HOCl oxidizes these phospholipids directly, disrupting the membrane’s structural integrity. Once the membrane is compromised, the cell loses its ability to regulate what enters and exits, and it rapidly lyses — essentially bursting from osmotic pressure.
2. Enzyme Inactivation
Inside the microbial cell, enzymes drive every metabolic process: energy production, respiration, protein synthesis. Many of these enzymes contain sulfhydryl groups — chemical bonds that HOCl reacts with immediately upon contact. When these bonds are disrupted, the enzyme stops functioning. The cell can no longer carry out basic metabolic activity and effectively shuts down.
3. DNA Damage
HOCl oxidizes the nucleic acids that make up microbial DNA. When the DNA is damaged in this way, the cell loses the ability to replicate or transcribe genetic information. Even if a pathogen somehow survived the membrane and enzyme attacks, it cannot reproduce — ending its ability to sustain or spread infection.
4. ATP Depletion
ATP (adenosine triphosphate) is the fundamental energy currency of all living cells. HOCl’s oxidative disruption of the cellular respiration pathways eliminates the cell’s ability to produce ATP within seconds. Without energy, every process the cell depends on stops. This pathway works in concert with the others, ensuring that inactivation is rapid and complete.
| The result of four simultaneous pathways:• Complete pathogen inactivation in under 30 seconds• Effective against bacteria, viruses, fungi, and spores• Disrupts even Staphylococcus aureus and Pseudomonas aeruginosa biofilms• No documented development of resistance — unlike antibiotic-based approaches• Multi-target action makes it effective against antibiotic-resistant organisms |
COMPARATIVE ANALYSIS
HOCl vs. Conventional Antiseptics
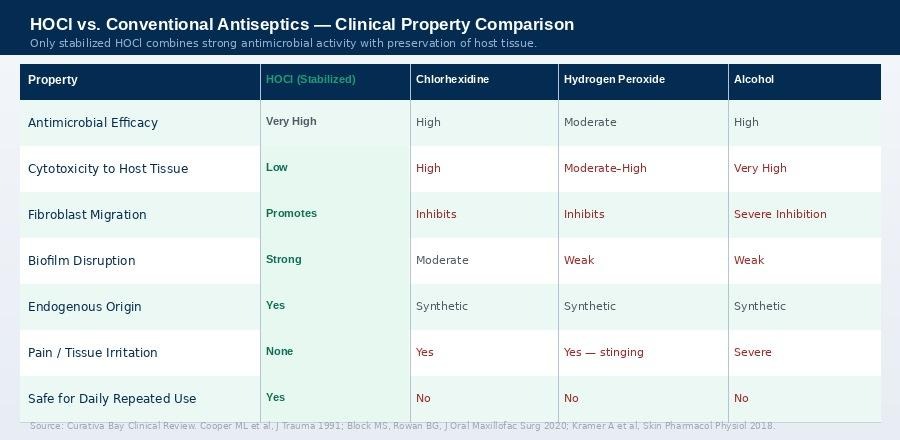
Clinical property comparison: HOCl (stabilized) versus chlorhexidine, hydrogen peroxide, and alcohol. Source: Curativa Bay Clinical Review; Cooper ML et al., J Trauma 1991; Kramer A et al., Skin Pharmacol Physiol 2018.
The comparison above reflects a fundamental tension in wound care that has existed for decades: the most effective antimicrobial agents tend to be the most harmful to host tissue. Alcohol is highly effective — it is also cytotoxic in a way that severely inhibits fibroblast migration and tissue repair. Hydrogen peroxide kills bacteria and also damages the healthy cells trying to heal the wound.
Chlorhexidine, one of the most widely used antiseptics globally, presents the same problem. It has strong antimicrobial activity and it inhibits fibroblast migration — meaning it actively impairs the cellular process by which wounds close. Studies have shown it to be cytotoxic to the keratinocytes and endothelial cells that healthy wound healing depends on.
Stabilized HOCl is the only agent in this comparison that achieves high antimicrobial efficacy while simultaneously promoting — not merely tolerating, but actively promoting — fibroblast migration and the cellular processes required for tissue regeneration.
| Conventional antiseptics were designed for sterilization, not regeneration. HOCl is the first clinically validated agent that achieves both in the same molecule. |
HEALING BIOLOGY
Supporting the Cellular Machinery of Wound Repair
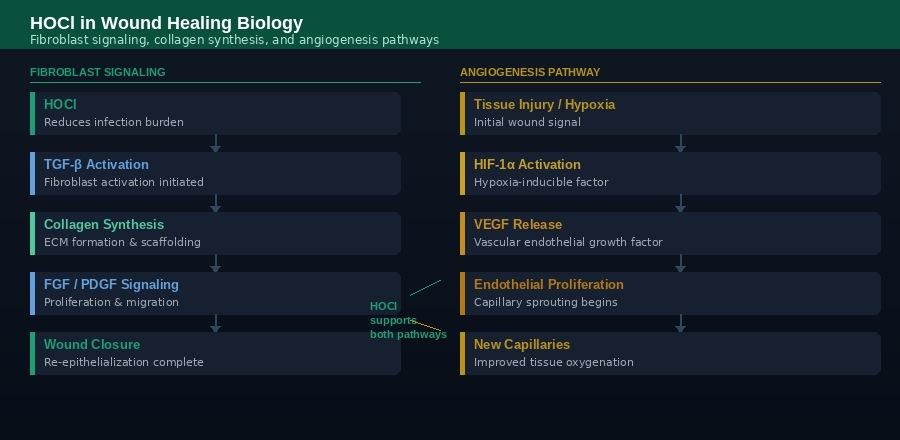
HOCl supports both fibroblast signaling pathways (collagen synthesis and wound closure) and angiogenesis (new blood vessel formation) — the two critical cellular tracks required for tissue regeneration.
Understanding why HOCl helps wounds heal requires a brief look at what wound healing actually involves at the cellular level. It is not simply a matter of keeping an area clean — healing is an active, highly coordinated biological process involving multiple cell types, signaling molecules, and vascular events.
Fibroblast Signaling: Building New Tissue
Fibroblasts are the cells responsible for synthesizing collagen, elastin, and the extracellular matrix proteins that give new tissue its structure and strength. Their activity is governed by a cascade of growth factor signals: TGF-β activates fibroblasts and initiates collagen synthesis; FGF drives proliferation and tissue expansion; PDGF guides migration into the wound bed to support closure.
Conventional antiseptics — particularly povidone-iodine, chlorhexidine, and hydrogen peroxide — disrupt these signaling pathways and inhibit fibroblast viability and migration. Stabilized HOCl, in dose-dependent studies, has been shown to preserve fibroblast viability across all three signaling pathways. It reduces the infection burden that would otherwise impair these pathways without causing the collateral damage that conventional agents produce.
Angiogenesis: Growing the Blood Supply
A wound cannot heal without adequate blood supply. New capillary growth — angiogenesis — is essential for delivering oxygen and nutrients to healing tissue. The process begins when tissue injury and hypoxia activate HIF-1α (hypoxia-inducible factor), which triggers VEGF (vascular endothelial growth factor) release. VEGF drives endothelial cell proliferation and capillary sprouting into the wound bed.
High bacterial load and biofilm presence suppress angiogenesis by maintaining a chronic inflammatory state that prevents the signal cascade from completing. HOCl supports angiogenesis by eliminating this suppressive microbial burden, modulating NF-κB inflammatory signaling, and maintaining the endothelial cell function required for capillary sprouting. Clinical evidence shows improved granulation tissue formation and vascularization in HOCl-treated wounds compared to controls.
BIOFILM DISRUPTION
The Hidden Enemy in Chronic Wounds
Biofilms are among the most significant barriers to wound healing that clinicians encounter, particularly in chronic wounds. A biofilm is a structured community of microorganisms — often including multiple species — encased in a self-produced matrix of polysaccharides, proteins, and DNA. This matrix functions as a physical shield that dramatically reduces the penetration and effectiveness of most conventional antibiotics and antiseptics.
Estimates suggest that biofilms are present in more than 60% of chronic wounds. Staphylococcus aureus and Pseudomonas aeruginosa are among the most common biofilm-forming organisms in clinical wound settings. Standard antibiotics may require concentrations 100 to 1,000 times higher than normal minimum inhibitory concentrations to penetrate established biofilm matrix.
HOCl disrupts biofilm through its high ORP activity, which oxidizes the polysaccharide matrix itself — not merely the organisms inside it. Laboratory studies published in Scientific Reports (Kiamco et al., 2019) demonstrated that HOCl-generating scaffolds eradicated established bacterial biofilms, including mature biofilms that had resisted conventional antiseptic treatment.
| HOCl and biofilm: what the evidence shows:• Oxidizes and degrades the extracellular polysaccharide matrix• Eliminates multi-species biofilms including MRSA and Pseudomonas aeruginosa• Does not create the conditions for biofilm re-establishment (no chemical residue)• Compatible with biofilm debridement protocols as an irrigation agent |
CLINICAL EVIDENCE
What the Research Demonstrates Across Wound Types

HOCl clinical evidence spans diabetic foot ulcers (randomized controlled trials), burn wounds (irrigation studies), and chronic leg wounds (first-in-human studies). Reference list: www.curativabay.com/clinical
The clinical case for HOCl in wound care is not built on a single study. It rests on a growing body of evidence across wound types, patient populations, and clinical settings — with 50 peer-reviewed references supporting the Curativa Bay clinical review.
Diabetic Foot Ulcers
Diabetic foot ulcers represent one of the most challenging wound types in clinical practice. Impaired immune function, peripheral neuropathy, and microvascular disease create conditions where infection is difficult to control and healing is chronically delayed. Randomized clinical trials evaluating HOCl in diabetic foot ulcer management have demonstrated statistically significant improvements in infection control outcomes, reductions in purulent exudate, lower bacterial burden on wound surfaces, and faster rates of wound closure compared to conventional antiseptic regimens. Multiple ongoing randomized controlled trials are currently enrolling hundreds of participants specifically to evaluate HOCl outcomes in this population.
Burn Wound Irrigation
Burn wounds present a different challenge: the wound surface is large, the risk of colonization is high, and the tissue environment is acutely fragile. Clinical studies evaluating HOCl as a burn wound irrigation agent have consistently shown meaningful advantages over conventional antiseptics, including reductions in bacterial burden, decreased wound odor (a marker of anaerobic bacterial activity), lower reported pain levels during irrigation — a clinically significant advantage given that conventional antiseptics often cause significant stinging and discomfort — and accelerated tissue healing rates.
Chronic Leg Wounds
First-in-human clinical studies evaluating HOCl in chronic leg wounds — including venous leg ulcers — confirmed safe repeated application and demonstrated beneficial effects on healing trajectories. Wound bed preparation improved, with evidence of reduced biofilm burden and improved granulation tissue formation. Researchers noted that HOCl was compatible with all wound types evaluated and produced no adverse tissue reactions across the patient population studied.
CONCLUSION
A Paradigm Shift in Wound Care
The history of wound antisepsis has been a story of trade-offs. Every conventional agent available has provided some benefit while also creating some harm to the tissue environment it is supposed to support. The clinical consensus has tolerated this trade-off because there was no alternative.
Stabilized HOCl changes that calculus. It is the only clinically validated wound care agent that combines high oxidation-reduction potential antimicrobial activity with active support for the biological processes required for tissue regeneration: fibroblast signaling, angiogenesis, matrix formation, and re-epithelialization.
It does this because it is not a foreign chemical introduced to the body. It is a molecule the body already produces, already understands, and already uses as its first response to tissue injury and infection. Stabilizing it for external clinical use is not a subversion of biology. It is an extension of it.
| No conventional antiseptic achieves what stabilized HOCl achieves: strong antimicrobial activity, biofilm disruption, and active support for the cellular biology of healing — simultaneously, in the same molecule. |
| 🦠Rapidly eliminates pathogens across all classes | 🔬Disrupts biofilm at the matrix level | 🧫Preserves fibroblast viability and migration | ⚡Accelerates wound closure and re-epithelialization |
Key References (Selected from 50 Cited)
1. Winter GD. Formation of scab and epithelialization. Nature. 1962;193:293.
2. Robson MC, et al. Hypochlorous acid as wound care agent: Part II. J Burns Wounds. 2007;6:e6.
3. Wang L, et al. Hypochlorous acid as wound care agent: Part I. J Burns Wounds. 2007;6:e5.
4. Winterbourn CC, Kettle AJ. Redox reactions in neutrophil phagosome. Antioxid Redox Signal. 2013;18:642.
5. Cooper ML, et al. Cytotoxic effects of topical antimicrobial agents on fibroblasts. J Trauma. 1991;31:775.
6. Kiamco MM, et al. HOCl-generating scaffold eradicates bacterial biofilms. Sci Rep. 2019;9:2683.
7. Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461.
8. Martínez-De Jesús FR, et al. Efficacy of super-oxidized solution in diabetic foot. Int Wound J. 2007;4:353.
9. Barrientos S, et al. Growth factors in wound healing. Wound Repair Regen. 2008;16:585.
10. Kramer A, et al. Consensus on wound antisepsis. Skin Pharmacol Physiol. 2018;31:28.
Full reference list: www.curativabay.com/clinical
| Curativa Bay HOCl Wound CareClinical-grade stabilized HOCl · EPA Certified · FDA Recognized · WHO CitedORP 850–1,100 mV · 80–100× more effective than bleach · Made in the USA · Since 2016www.curativabay.com · www.curacleantech.com |
Copyright © 2026 Curativa Bay, Inc. · Curaclean Technologies. All rights reserved. This article is for educational purposes. It is not intended as medical advice. Consult a qualified healthcare provider for clinical wound management.